
For research use only and NOT intended for in vitro diagnosis.
Pli-RCA TB RIF/INH LF
For the qRCA detection of M. tuberculosis complex and associated drug resistance genes to rifampicin and isoniazid.
Key features
- Validated with clinical samples
- 15 gene targets
- Platform agnostic
- Tolerant of blood contaminants
- Manufactured in ISO 13485 certified facilities
Pack size
- 25 Reactions
×
EMPE TBDx (RUO) Launch Celebration Offer
To celebrate the launch, enjoy our exclusive promotion:
Buy 2 kits for the price of 1.Offer valid until 30th April 2026!
Order Now!Description
EMPE Diagnostics, deploying its core Pli-RCA technology (Padlock probe–linked Rolling Circle Amplification) coupled with the patented mfloDx™ visualization platform, enables for the easy identification of the presence of TB and its associated drug resistance genes.
This integrated approach enables the rapid, accurate, and cost-effective detection of Mycobacterium tuberculosis complex (MTBC) and its 15 genes distinguishing between sensitive and resistant strains to rifampicin (RIF) and isoniazid (INH) directly from purified genomic DNA, extracted from clinical (smear positive) or non-clinical samples, in under three hours.
The detection signal is processed and visualized using a lateral flow cassette. “Wild type” (WT) and “mutant” (MUT) windows indicate the presence of the bacterium and its specific drug-resistance profile.
The Pli-RCA TB RIF/INH LF is for research use only and NOT intended for in vitro diagnosis.
This integrated approach enables the rapid, accurate, and cost-effective detection of Mycobacterium tuberculosis complex (MTBC) and its 15 genes distinguishing between sensitive and resistant strains to rifampicin (RIF) and isoniazid (INH) directly from purified genomic DNA, extracted from clinical (smear positive) or non-clinical samples, in under three hours.
The detection signal is processed and visualized using a lateral flow cassette. “Wild type” (WT) and “mutant” (MUT) windows indicate the presence of the bacterium and its specific drug-resistance profile.
The Pli-RCA TB RIF/INH LF is for research use only and NOT intended for in vitro diagnosis.
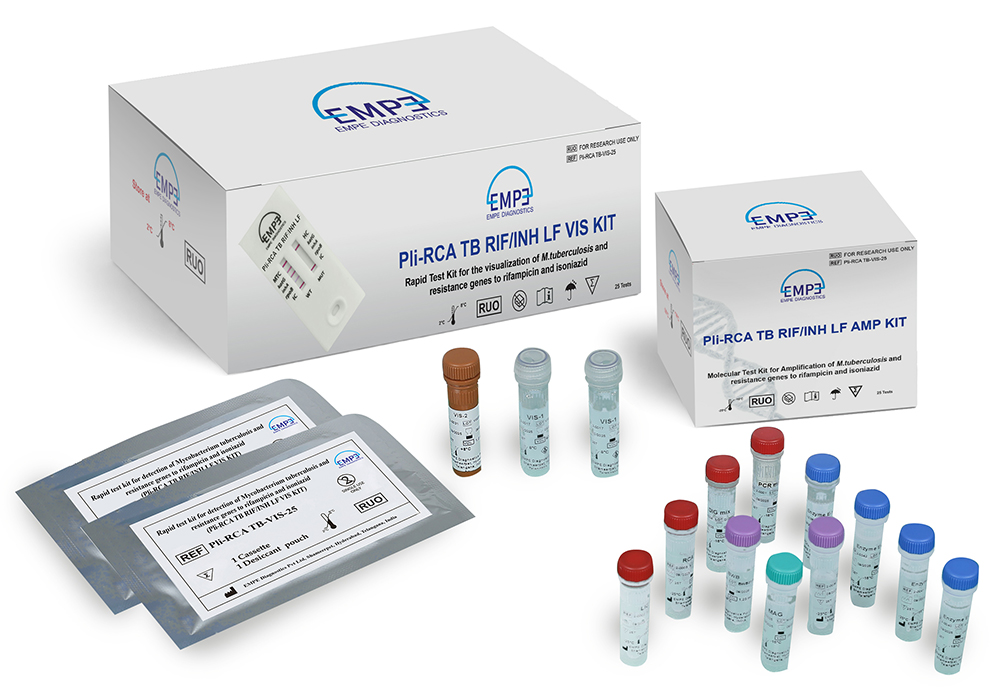
Kit contents
The AMP Kit contents supplied with the Pli-RCA TB RIF/INH LF AMP Kit as detailed below:
The VIS Kit contents supplied with the Pli-RCA TB RIF/INH LF VIS Kit as detailed below:
| Name of the Product | Quantity supplied with the kit |
|---|---|
| PCR mix | 1 x 360.0 µl |
| LIG mix | 1 x 450.0 µl |
| RCA mix | 1 x 480.0 µl |
| DIG mix | 1 x 112.5 µl |
| Enzyme E-1 | 1 x 9.0 µl |
| Enzyme E-2 | 1 x 25.0 µl |
| Enzyme E-3 | 1 x 20.0 µl |
| Enzyme E-4 | 1 x 12.5 µl |
| MAG | 1 x 75.0 µl |
| BWB | 2 x 1.25 ml |
The VIS Kit contents supplied with the Pli-RCA TB RIF/INH LF VIS Kit as detailed below:
| Name of the Product | Quantity Supplied with the Kit |
|---|---|
| VIS-1 | 2 x 937.5 µl |
| VIS-2 | 1 x 1.0 ml |
| Cassettes | 1 x 25 units |
Storage and handling
Storage and Handling (AMP)
Storage and Handling (VIS)
The Pli-RCA TB RIF/INH LF AMP Kit should be stored at -25 °C to -16 °C.
The number of freeze-thaw cycles with the 25-test kit is 4. Beyond 4 times, the kit cannot be used and the results obtained beyond the 4th cycle may not be considered for interpretation.
Storage and Handling (VIS)
The Pli-RCA TB RIF/INH LF AMP Kit should be stored at +2 °C to +8 °C. The number of temperature excursions for the 25-test kit is 4, based on the AMP kit freeze-thaw studies. Beyond 4 times, the kit cannot be used and the results obtained beyond the 4th cycle may not be considered for interpretation.


