
For in vitro diagnostic use.
mfloDx™ MDR-TB
Molecular test kit to aid in the diagnosis of Mycobacterium tuberculosis complex (MTBC) infection and detection of resistance to rifampicin and isoniazid.
Key features
- Validated with clinical samples including extrapulmonary
- Peer-reviewed performance studies
- Platform agnostic
- Tolerant of blood contaminants
- CDSCO-approved for IVD use (Indian regulatory authorities)
Pack size
- 25 reactions
Description
EMPE Diagnostics, deploying its core Pli-RCA technology (Padlock probe–linked Rolling Circle Amplification) coupled with the patented mfloDx™ visualization platform, enables for the easy identification of the presence of TB and its associated drug resistance genes in diagnostic settings.
This integrated approach enables the rapid, accurate, and cost-effective detection of Mycobacterium tuberculosis complex (MTBC) and its resistance genes to rifampicin (RIF) and isoniazid (INH) directly from sputum samples within three hours.
The detection signal is processed and visualized using a lateral flow cassette. “Wild type” (WT) and “mutant” (MUT) windows indicate the presence of the bacterium and its specific drug-resistance profile.
The technology provides clinicians with a qualitative “Yes” or “No” result for both TB presence and drug resistance, enabling the prompt initiation of appropriate treatment, particularly in resource-limited settings.
The mfloDx™ MDR-TB test has been cleared for IVD use in India by the Central Drugs Standard Control Organization (CDSCO).
This integrated approach enables the rapid, accurate, and cost-effective detection of Mycobacterium tuberculosis complex (MTBC) and its resistance genes to rifampicin (RIF) and isoniazid (INH) directly from sputum samples within three hours.
The detection signal is processed and visualized using a lateral flow cassette. “Wild type” (WT) and “mutant” (MUT) windows indicate the presence of the bacterium and its specific drug-resistance profile.
The technology provides clinicians with a qualitative “Yes” or “No” result for both TB presence and drug resistance, enabling the prompt initiation of appropriate treatment, particularly in resource-limited settings.
The mfloDx™ MDR-TB test has been cleared for IVD use in India by the Central Drugs Standard Control Organization (CDSCO).
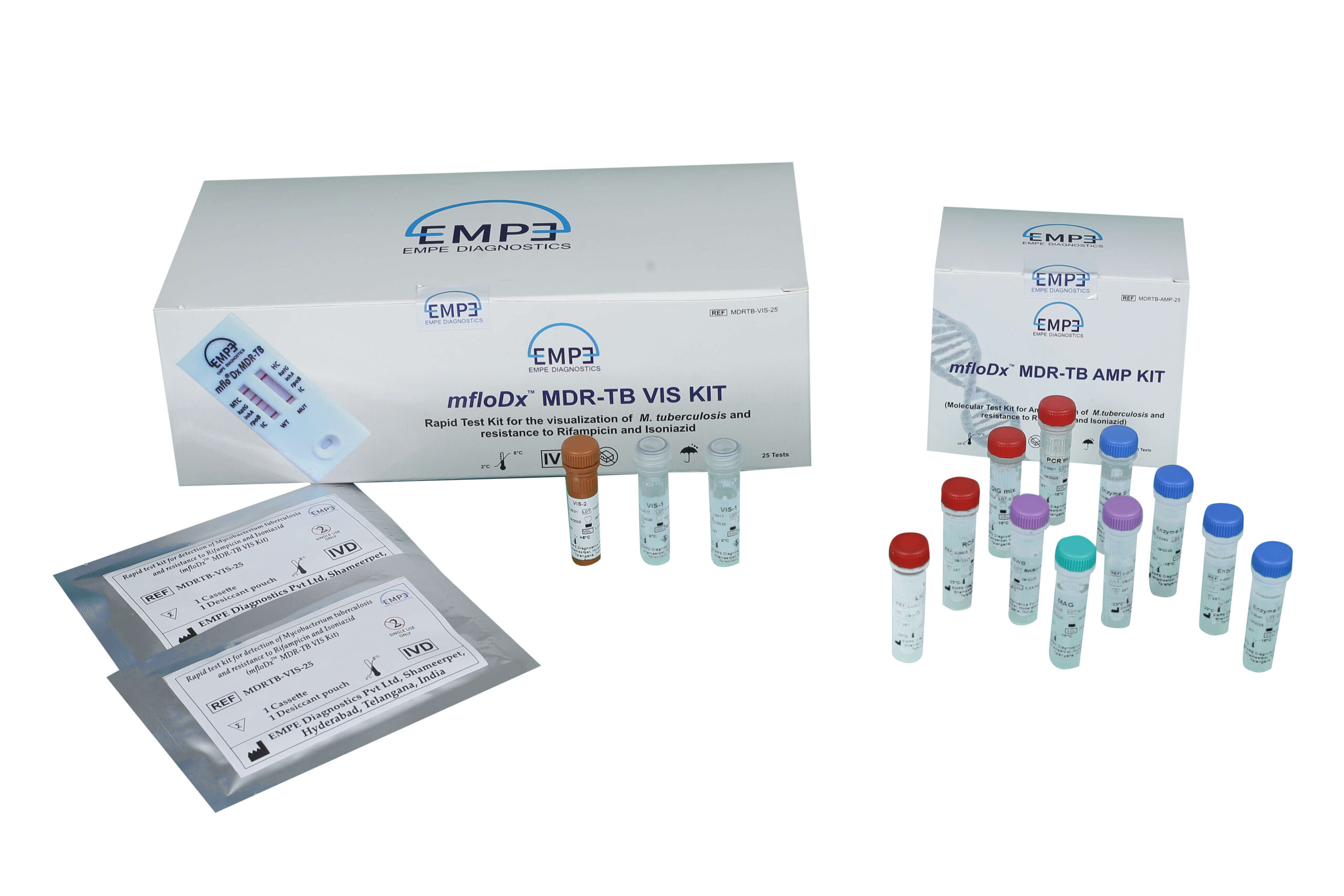
Kit contents
The AMP Kit contents supplied with the mfloDx™ MDR-TB AMP Kit as detailed below:
The VIS Kit contents supplied with the mfloDx™ MDR-TB VIS Kit as detailed below:
| Name of the Product | Quantity supplied with the kit |
|---|---|
| PCR mix | 1 x 360.0 µl |
| LIG mix | 1 x 450.0 µl |
| RCA mix | 1 x 480.0 µl |
| DIG mix | 1 x 112.5 µl |
| Enzyme E-1 | 1 x 9.0 µl |
| Enzyme E-2 | 1 x 25.0 µl |
| Enzyme E-3 | 1 x 20.0 µl |
| Enzyme E-4 | 1 x 12.5 µl |
| MAG | 1 x 75.0 µl |
| BWB | 2 x 1.25 ml |
The VIS Kit contents supplied with the mfloDx™ MDR-TB VIS Kit as detailed below:
| Name of the Product | Quantity Supplied with the Kit |
|---|---|
| VIS-1 | 2 x 937.5 µl |
| VIS-2 | 1 x 1.0 ml |
| Cassettes | 1 x 25 No. |
Storage and handling
Storage and Handling (AMP)
Storage and Handling (VIS)
The mfloDx™ MDR-TB AMP Kit should be stored at -25 °C to -16 °C. The number of freeze-thaw cycles with the 25-test kit is 4. Beyond 4 times, the kit cannot be used and the results obtained beyond the 4th cycle may not be considered for interpretation.
Storage and Handling (VIS)
The mfloDx™ MDR-TB VIS Kit should be stored at +2 °C to +8 °C.
The number of temperature excursions for the 25-test kit is 4, based on the AMP kit freeze-thaw studies. Beyond 4 times, the kit cannot be used and the results obtained beyond the 4th cycle may not be considered for interpretation.


